CO2-electrolysis promises to convert harmful CO2 into usable chemicals such as carbon monoxide (CO) or ethylene glycol with the help of renewable energy. These, in turn, can serve as feedstock chemicals for alcohols, synthetic fuels or plastics and are thus key to “de-fossilising” the chemical industry.
Precipitates slow down efficiency
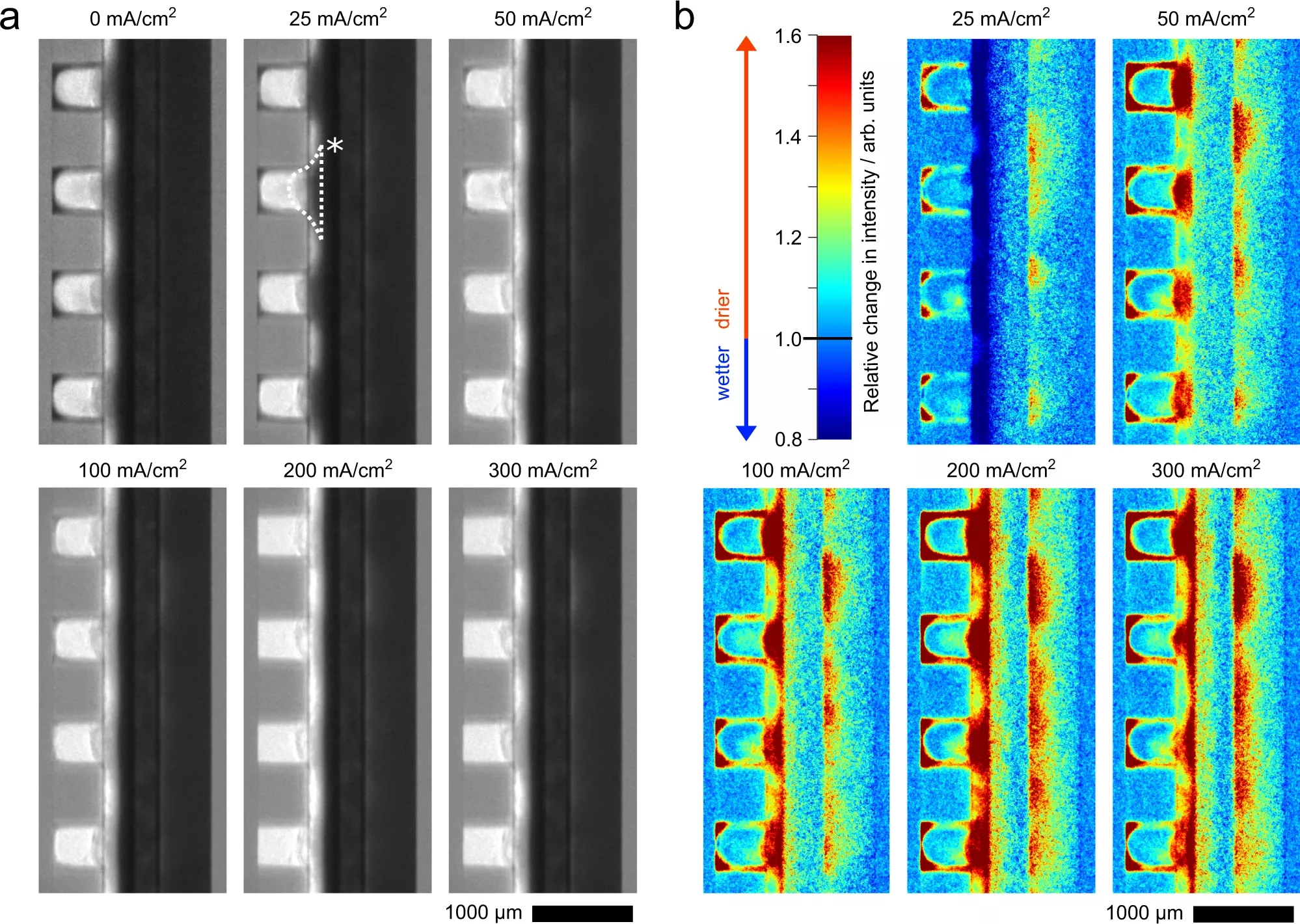
In CO2-electrolysis, analogous to water electrolysis, i.e. hydrogen production, electrochemical reactions are carried out at two electrodes separated by a membrane. This continuously converts CO2 into CO. The development can therefore be based on previous experience with water electrolysis and with the fuel cell – i.e. the conversion of hydrogen back into electricity, e.g. to power a truck. This is because the cathode electrode is designed as a so-called gas diffusion electrode, similar to the fuel cell. Unlike in the fuel cell, however, in CO2-electrolysis gas diffusion can be impeded by the formation of salts. These salts are formed as an unwanted by-product and prevent the CO2 from reaching the catalyst and being converted into CO. This salt formation was already known, but it has not yet been detected in operation in the cell.
Neutrons make salts visible for the first time
In a cooperation with the Research Neutron Source Heinz Maier-Leibnitz of Technical University of Munich (TUM), the CNRS and Institut Laue-Langevin in Grenoble, the research group “Electrochemical Energy Systems” at the University of Freiburg has now been able to show for the first time in high resolution how salt formation takes place in the electrode. This means that further steps can now be taken to prevent salt formation and thus achieve consistently high efficiency.
International cooperation
The study, published in the journal Nature Communications, can be seen as a prime example of European interdisciplinary cooperation: while the electrochemical experiments were developed at the University of Freiburg, scientists from TUM contributed a neutron detector with record resolution. Finally, all the building blocks were put together in Grenoble, because that is where the CO2-electrolysis experiments were set up at the ILL neutron source and measured with the Munich detector.
Original text: University of Freiburg
Original publication:
Joey Disch, Luca Bohn, Susanne Koch, Michael Schulz, Yiyong Han, Alessandro Tengattini, Lukas Helfen, Matthias Breitwieser, Severin Vierrath
High-resolution neutron imaging of salt precipitation and water transport in zero-gap CO2 electrolysis
Nature Communications 13, 6099 (2022)
Contact
Dr. Severin Vierrath
Head of the Junior Research Group „Electrochemical Energy Systems”
IMTEK – Department of Microsystems Engineering
University of Freiburg
FIT – Freiburg Center for Interactive Materials and Bioinspired Technologies
Phone: +49 761 203 54060
Email: severin.vierrath(at)imtek.uni-freiburg.de
Web : www.ees-lab.org
Dr. Michael Schulz
Head of Neutron Imaging Group
Technical University of Munich
Forschungs-Neutronenquelle
Heinz Maier-Leibnitz (FRM II)
Lichtenbergstr. 1
85748 Garching
Germany
Phone: +49 (0)89 289-14718
Email: michael.schulz(at)frm2.tum.de